A rigid diatomic ideal gas undergoes an adiabatic process at room temperature. The relation between temperature and volume of this process is TVx 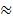 constant, then x is :
constant, then x is :
(a)

(b)

(c)

(d)

A rigid diatomic ideal gas undergoes an adiabatic process at room temperature. The relation between temperature and volume of this process is TVx 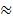 constant, then x is :
constant, then x is :
(a)

(b)

(c)

(d)

Question ID - 50300 | SaraNextGen Answer
A rigid diatomic ideal gas undergoes an adiabatic process at room temperature. The relation between temperature and volume of this process is TVx 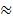 constant, then x is :
constant, then x is :
(a)

(b)

(c)

(d)

|
A rigid diatomic ideal gas undergoes an adiabatic process at room temperature. The relation between temperature and volume of this process is TVx |
|||
|
(a) |
|
(b) |
|
|
(c) |
|
(d) |
|
1 Answer - 5876 Votes
3537
Answer Key : (b) -
Answer LinkFor adiabatic process : TV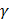 −1=constant
−1=constant
For diatomic process :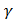 −1 =
−1 =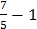
∴x=
